[Funding News] MD-based Amplexd Therapeutics Secures USD2Million in Funding
May 20, 2024 | By Startuprise io
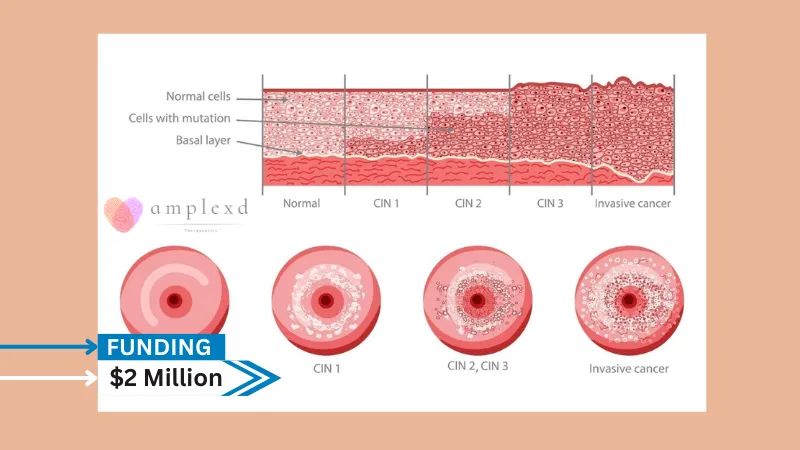
Amplexd Therapeutics, Inc. (“Amplexd” or the “Company”), a clinical stage patient-scientist-led biotech company specializing in accessible treatments for human papillomavirus (HPV)-induced Cervical Intraepithelial Neoplasia (CIN), is pleased to announce major updates in 2024 toward the finalization of product development ahead of Phase 1/2 clinical trials for its non-invasive treatments of CIN, the precursor to cervical cancer. The Company secures USD2million in funding for this initiative from an Asia-based life sciences focused family office.
Co-Founder and CEO Alia Rahman said, “The funding marks a major milestone in our R&D efforts for our two therapies after extremely promising preclinical studies were completed. The capital will enable us to finalize development and IND submissions ahead of first-in-human clinical trials, expected to commence later this year,”.
The strategic funding positions Amplexd Therapeutics to reshape the landscape in HPV and cervical precancer treatments with two topical, non-invasive options as potential alternatives to the “watch and wait” approach for low-grade CIN, and invasive surgery for high-grade CIN.
RECOMMENDED FOR YOU

Oracle set to raise up to $50 billion in debt and equity this year
Startuprise io
Feb 2, 2026

[Funding News] Ascend Partners Secures Funding from Achieve Partners
Startuprise
Oct 8, 2024
Amplexd is poised to move the needle with the development of two low-cost therapies that favor patient comfort and increase access, and a vision to provide accessible solutions to target the often-overlooked public health crisis of HPV-induced cervical precancers.
Founded in 2022, Amplexd Therapeutics has been gaining prominence in the women’s health space as a company at the technological vanguard of the treatment of CIN, a disease which affects upwards of 178 million women globally every year.
Read also - [Funding News] CA-based Kudos Secures $10.2Million in Series A Round Funding
About 5% of cervical screenings in the US and Europe reveal abnormalities at any given time, with significantly higher rates in Asia, Latin America, and Africa. CIN results from certain oncogenic varieties of HPV, which can transform cervical cells into invasive cancer if left untreated. Cervical cancer is the fourth leading cause of cancer death globally in women and can largely be prevented by treatment at the CIN stage.1
About Amplexd Therapeutics, Inc.
Amplexd Therapeutics is developing two low-cost, novel, non-invasive therapies for the treatment of Cervical Intraepithelial Neoplasia (cervical precancer). The first is an intravaginal suppository for low-grade CIN, and the second is a photodynamic therapy (PDT) system for high-grade CIN, each designed to selectively target neoplastic cells.



 Follow us
Follow us Follow us
Follow us













